Chemical bonds

Description
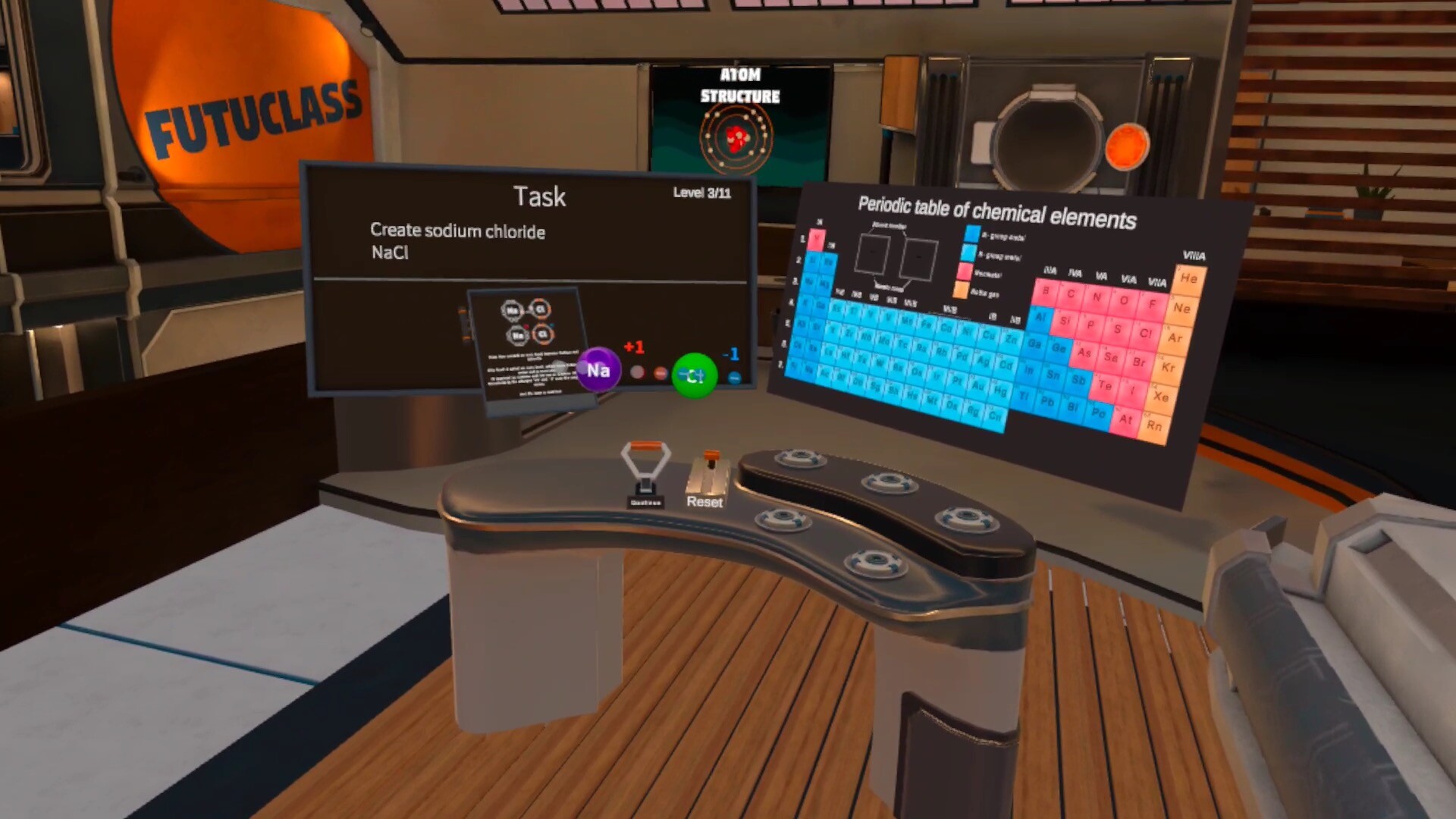
This module will give the student essential knowledge about the principles of chemical bond creation. Ionic and covalent bonds are covered.
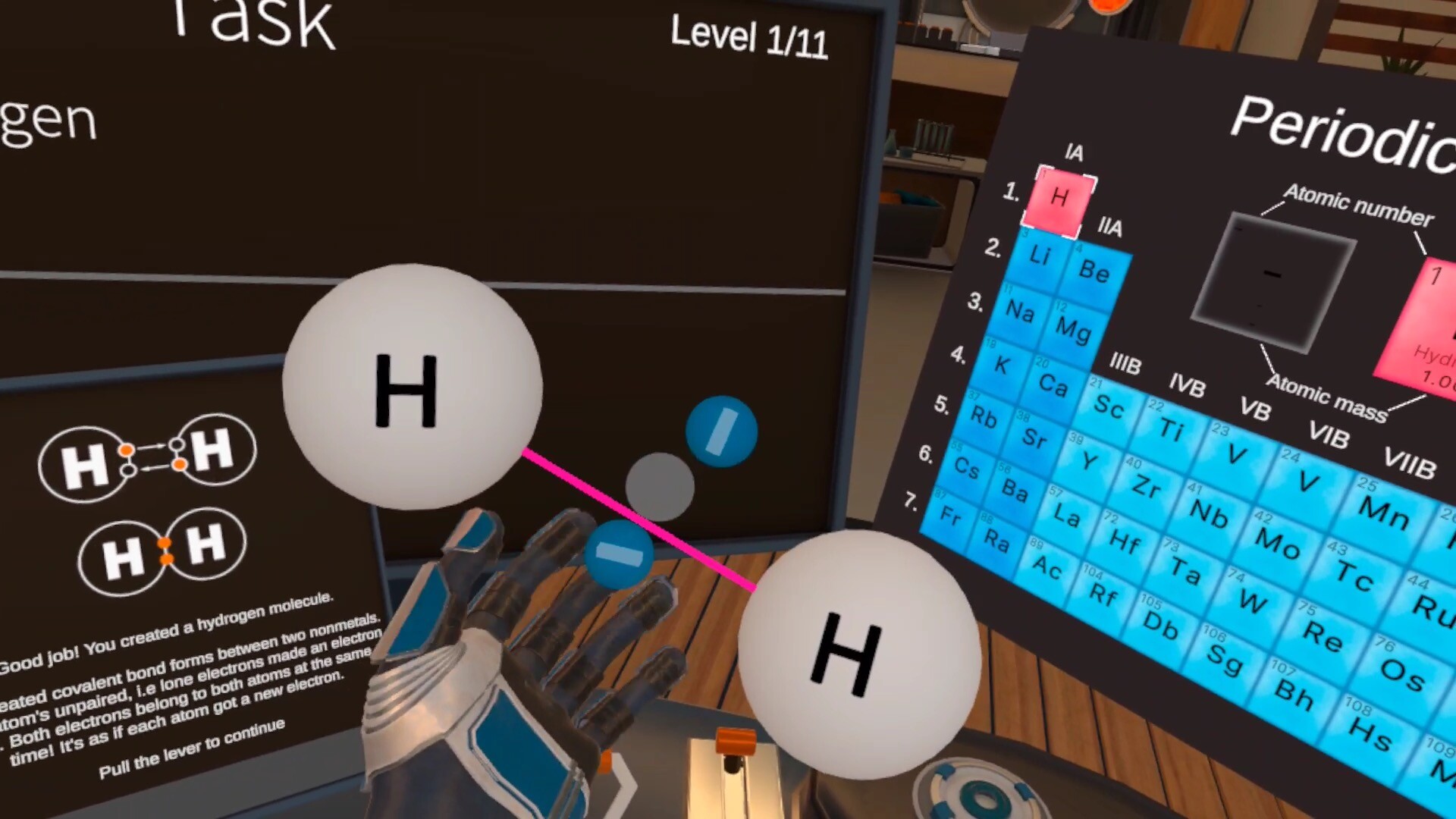
The student will create both types of chemical bonds, and decide the bond type based on the substance classes involved.
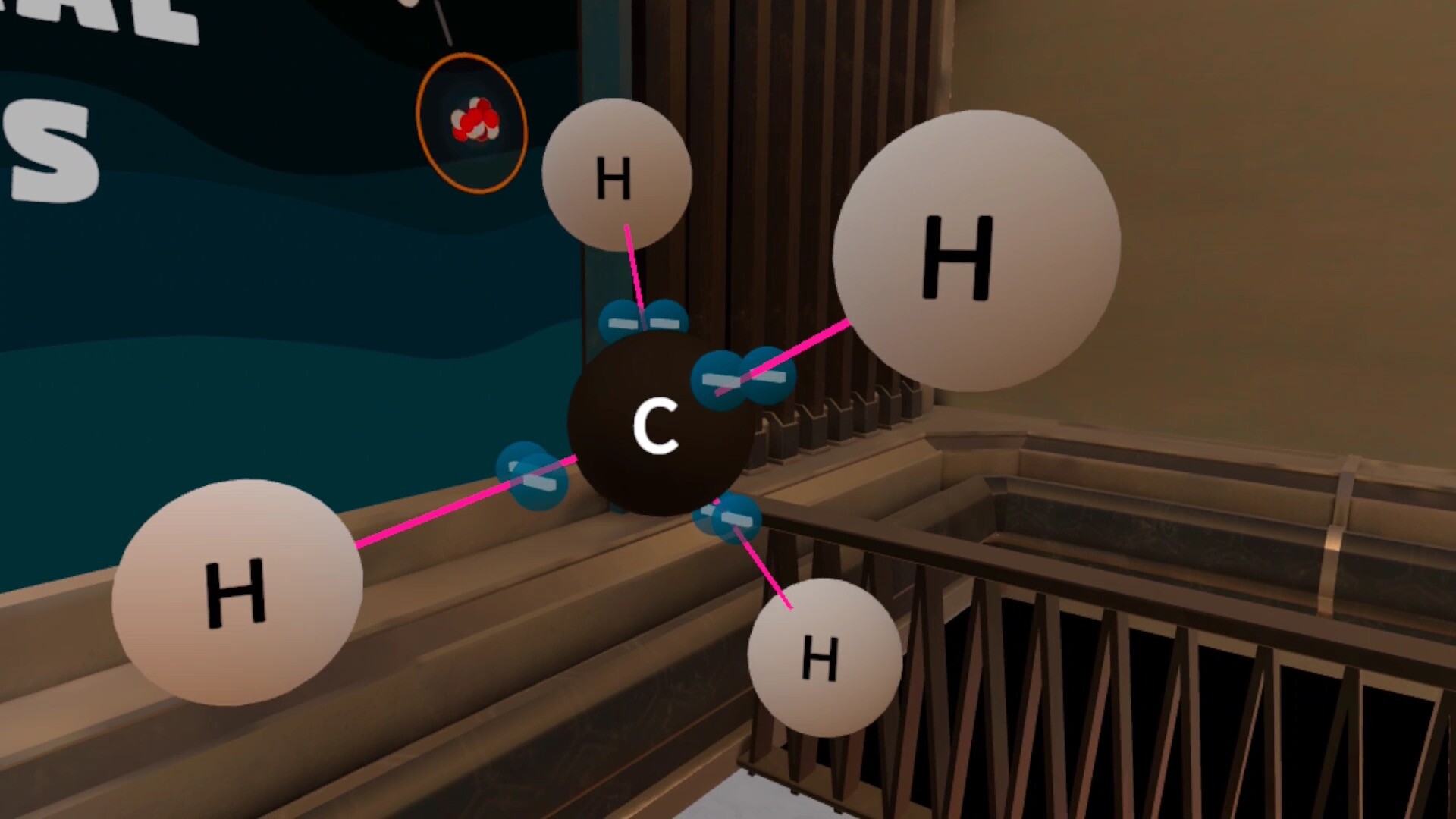
Student will learn about: valence electrons, metals and nonmetals, ionic bond, covalent bond, octet rule, lone electrons, double bond, triple bond.
Prerequsites
Completion of the Atom Structure VR lesson. Can draw an electron diagram of an atom. Familiar with the octet rule. Familiar with the periodic table. Knows the classification of chemical elements into metallic and non-metallic and their location in the periodic table.
Learning objectives
- Describe the difference between a covalent and an ionic bond
- Distinguish ions from neutral atoms and can explain the formation of ions and the charge of an ion
- Understand the difference between molecular (made up of molecules) and non-molecular substances, and give examples of each.